Current Events
FDA Approves CBD Treatment for Children
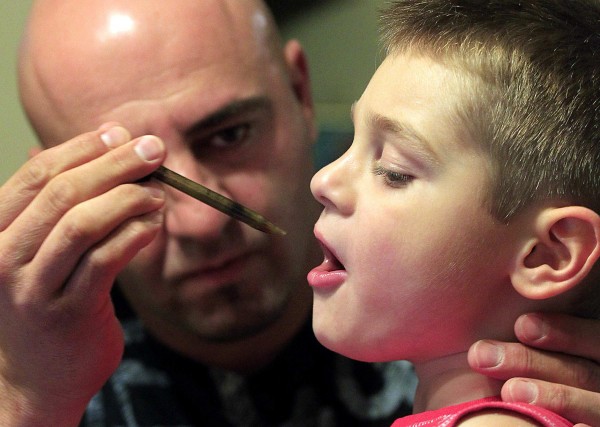
Jason David administers oral drops of a medical marijuana tincture that he says has greatly helped control the symptoms of his son Jayden’s severe epilepsy in Modesto, California, July 20, 2012.
photo: Luis Sinco | Los Angeles Times
Children with Rare Epilepsy to be Treated with Cannabis
A drug made with cannabis has been approved by the FDA to treat children with rare epileptic conditions. GW Pharmaceuticals has announced FDA orphan drug designation for Epidiolex, a drug that contains plant-derived Cannabidiol (CBD), for treating 125 children. Orphan drug designation, approved for rare conditions that generally affect fewer than 200,000 individuals, means the U.K.-based biopharmaceutical company will receive tax incentives as well as a seven-year marketing monopoly on the drug.
CBD, a compound in cannabis that does not produce the psychoactive affects of Tetrahydrocannabinol (THC), has been shown to treat a number of conditions including Dravet syndrome. The syndrome, highlighted in the CNN documentary “Weed” by Dr. Sanjay Gupta earlier this year, is a rare form of childhood epilepsy. In Gupta’s documentary CBD is a lifeline for Charlotte Figi, a 6-year-old who suffers from the syndrome that makes her prone to up to 300 seizures per week.
GW Pharmaceuticals also produces Sativex, an oral spray that includes both CBD and THC and is approved for the treatment of multiple sclerosis in Canada, New Zealand, Spain and the U.K.
Tell Us in the comments below, do you believe children should be allowed to be treated with medical marijuana?





















